Thermal diffusivity is a measure of the transient thermal response of a material to a change in temperature and the term thermal diffusivity (α) is defined asα= k/(ρ x cp)
| where | αis the thermal diffusivity (m2/sec) |
| k is the thermal conductivity (W/m-K) | |
| ρ is the density (kg/m3) | |
| cp is the heat capacity (J/kg-K) |
It should be noted that each of these quantities can vary with temperature. Thermal diffusivity is a convenient collection of physical properties for transient solutions of the heat equation. For a homogenous material with constant properties, the heat equation (1) with three physical properties is expressed as (2) with just one coefficient.
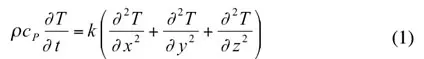 |
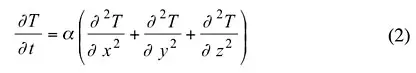 |
A material with a high thermal diffusivity (such as silver) is a good diffuser of thermal energy while a material with a low thermal diffusivity (such as plastic) is much slower at diffusing thermal energy. If the thermal environment around a material changes, heat must flow in or out of the material until thermal equilibrium is achieved, assuming the environment is constant after the change. Materials with a high thermal diffusivity will achieve thermal equilibrium faster than materials with low thermal diffusivity.
 |
Figure 1. Thermal conductivity versus thermal diffusivity for a wide variety of homogeneous materials (closed circles, metals; squares, ceramics; triangles, glasses; open squares, polymers; open circles, liquids; and crosses, gases) [1].
Figure 1 compares the thermal conductivity to thermal diffusivity for a wide range of materials. Notice that the data points noted as condensed matter can be described as being near a straight line. This is because the range of heat capacity per unit volume for condensed matter (liquids and solids) is small (the range of heat capacity per volume for condensed matter is from 1×10-6 J/m3 -K to 4×10-6 J/m3-K). With respect to thermal diffusivity, gases and condensed matter are different. For example, air has a low thermal conductivity but a relatively high thermal diffusivity � this means that while air can only absorb a relatively small amount of thermal energy, it is effective at diffusing the energy.
Thermal conductivity is a measure of the heat flow in a material resulting from a temperature difference. Obtaining accurate thermal conductivity measurements at steady-state conditions require knowing both the heat flux and the temperatures and additionally, that these conditions are non-changing in time. The time requirements and inherent difficulty in making accurate measurements has led to the development of transient techniques for property measurement. Heat capacity and density are relatively easy to measure and even predict given the constituent materials. They are also measurable on small sample sizes. A measurement of thermal diffusivity provides a means of extracting thermal conductivity. A thermal diffusivity measurement adds the need to measure time but accurate time measurement is not difficult. Measurement of thermal properties using a flash technique was first described in 1960 by Parker et al. of the U.S. Navy Radiological Defense Laboratory [2]. Further efforts have improved these measurement techniques to well established methods for material property measurements [3].
The units of thermal diffusivity are length2/time and a common set of SI units is m2/sec (the use of cm or mm as the length scale is frequent as it allows reporting of values closer to the value one). Table 1 lists thermal diffusivities for selected materials at room temperature (300 K). As with thermal conductivity, significant variation in reported values is common.
Table 1. Thermal Diffusivity for Selected Materials
|
References
- Salazar, A., “On Thermal Diffusivity,” European Journal of Physics, 24, 2003.
- Parker, W. et al., “A Flash Method of Determining Thermal Diffusivity, Heat Capacity, and Thermal Conductivity,” Journal of Applied Physics, 32, 1961.
- ASTM E1461-01 Standard Test Method for Thermal Diffusivity of Solids by the Flash Method, ASTM International, www.astm.org.
- King, J., Material Handbook for Hybrid Microelectronics, Artec House, Norwood, Mass., 1988.





